Funders:
- Bill and Melinda Gates Foundation
- The Wellcome Trust
- DFAT
- PATH
Collaborators:
- Asia Pacific Malaria Elimination Network (APMEN - Global)
- PATH (USA)
- Institute of Tropical Medicine (ITM - Belgium)
- FIND (Switzerland)
- International Centre for Diarrheal Diseases Research, Bangladesh (icddr,b - Bangladesh)
- Department of Health (Bhutan)
- National Institute of Parasitological Diseases (NIPD - China)
- Medical College Kalkutta (KMC - India)
- Eijkmans Institute of Molecular Biology (EIMB - Indonesia)
- Centre for Malaria Parasitology and Entomology (CMPE – Laos)
- Tribhuvan University (Nepal)
- Agha Khan University (AKU - Pakistan)
- National Institute of Malariology, Parasitology and Entomology (Vietnam)
Project team:
Chief investigators:
Menzies Investigators:
- Dr Jutta Marfurt
- Associate Professor Sarah Auburn
- Dr Kamala Ley-Thriemer
- Catherine Martel
Other Key invstigators:
- Dr Shafiul Alam (icddr,b, Bangladesh)
- Dr Khan (icddr,b, Bangladesh)
- Dr Sonam Wangchuk (Department of Health, Bhutan)
- Dr Wang (NIPD, China)
- Dr Talukdar (KMC, India)
- Dr Das (NICED, India)
- Dr Winasti-Satyagraha (EIMB, Indonesia)
- Dr Noviyanti (EIMB, Indonesia)
- Professor Ghimire (Tribhuavn University, Nepal)
- Professor Beg (AKU, Pakistan)
- Professor Ghanci (AKU, Pakistan)
- Dr Xa (NIMPE, Vietnam)
Project managers:
- Catherine Martel, Menzies
- Wulan Morling, Menzies
- The team at Agha Kahn University, Pakistan, has just enrolled their first patient.
Patients cuing at the field site in Karachi, Pakistan

- A workshop on a novel diagnostic to measure G6PD activity at the bed side was held in October 2019 in Bangladesh in collaboration with FIND, icddr,b, the University of Mastricht and Menzies.
Workshop participants in Bangladesh

-
Hidden malaria life cycle discovered in the spleen
Ground-breaking studies published today in the New England Journal of Medicine and PLOS Medicine have found large numbers of malaria parasites hiding in the human spleen where they actively multiply in a previously unrecognised life cycle.
-
Menzies investigators and international collaborators from ACROSS and other institutions have published a meta-analysis
Menzies investigators and international collaborators have published a meta-analysis and identified a point of care test for the diagnosis of G6PD deficiency with suitable reliability for routine use. Further feasibility studies are under way to assess its reliability under field conditions.
ACROSS Study
APMEN health Care facility & community assessment to determine populations at Risk Of malaria and primaquine-induced haemolysiS
Aim:
To identify populations and individuals at risk of malaria and drug induced hemolysis. Evaluating the utility of novel diagnostics.
Rationale:
In the Asia-Pacific region an increasing proportion of malaria is due to Plasmodium vivax since it is harder to eliminate than Plasmodium falciparum. P. vivax can form dormant liver stages that can relapse weeks to months after an initial infection. The only widely available drug to kill the liver stages is primaquine, but this can cause drug induced hemolysis in patients with a common enzyme deficiency known as Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency.
To promote the safe and effective use of primaquine radical cure for malaria, we need to have a better understanding of which populations are at greatest risk of malaria and know prevalence and variants of G6PD deficiency in these populations. In the last five years, great gains have been made in developing novel point-of-care diagnostics that can diagnose patients with P. vivax malaria and identify those with G6PD deficiency. The utility of these diagnostics in a clinical setting need to be explored both by quantitative and qualitative surveys. The aim of the ACROSS surveys is to provide a standardised approach to field testing so that a range of data can be gathered for each location, but can also be pooled for a comprehensive analysis.
Implications for policy and practice:
The study is designed to inform local policy makers on the local burden of malaria and G6PD deficiency, how well novel diagnostics perform in their country and identify key bottlenecks in patient and caregiver perceptions that may impede their widespread use.
For more information about the ACROSS Study, contact Dr Benedikt Ley.
Downloadable files:
- Generic study protocol
- Laboratory SOP
- Household and participants selection in cross sectional survey.
Novel diagnostics tested to date:
Malaria
- Autoscope: microscopy done by artificial intelligence
- us-LFA: a highly sensitive lateral flow assay for the diagnosis of P. falciparum at the bed side
G6PD deficiency
- STANDARD G6PD: A quantitative biosensor to measure G6PD activity and hemoglobin at the bed side
- Carestart G6PD RDT: A qualitative lateral flow assay to measure G6PD activity at the bed side
Publications:
- Ley, B., Rijal, K.R., Marfurt, J., Adhikari, N.R., Banjara, M.R., Shrestha, U.T., Thriemer, K., Price, R.N., & Ghimire, P. (2019). Analysis of erroneous data entries in paper based and electronic data collection. BMC Research Notes, 12, 537. https://doi.org/10.1186/s13104-019-4574-8.
- Phru, C.S., Kibria, M.G., Thriemer, K., Chowdhury, M.U., Jahan, N., Aktaruzzaman, M.M., Rahmat, H., Satyagraha, A.W., Prue, A.S., Khan, W.A., Ley, B., & Alam, M.S. (2019). Case Report: A Case of Primaquine-Induced Hemoglobinuria in Glucose-6-Phosphate Dehydrogenase-Deficient Malaria Patient in Southeastern Bangladesh. American Journal of Tropical Medicine and Hygiene. https://doi.org/10.4269/ajtmh.19-0643
Objectives:
Primary Objectives:
- To quantify the microscopic and sub microscopic prevalence of malaria
- To determine the prevalence and spectrum of activity of G6PD deficiency in patients with and without malaria.
Secondary Objectives:
- To evaluate novel qualitative and quantitative diagnostics for the diagnosis of G6PD deficiency
- To evaluate performance of currently used malaria RDTs
- To evaluate ultra-sensitive diagnostics for the diagnosis of malaria
- To identify utility of serological or other survey tools for targeting interventions, and appropriate ways to use them
- To evaluate the perceptions and utility of novel diagnostics in healthcare providers
- To determine the local G6PD and CYP2D6 genotypes present in the local population
- To determine the local prevalence of anemia
- To quantify prevalence of parasite polymorphisms associated with antimalarial drug resistance
- To assess community understanding of P. vivax infection and radical cure
- To assess community perceptions of P. vivax infection and the concept of radical cure
- To assess health seeking behavior for P. vivax malaria at community level
- To assess acceptability of G6PD testing among health system stakeholders, health facility staff and local communities
- To review prescription behavior for radical cure among health facility staff
- To assess adherence to radical cure prescription at health facility level and adherence to radical cure treatment at community level.
A training workshop with ACROSS collaborators in Vietnam facilitated by the Institute of Tropical Medicine, Antwerp, Belgium and Menzies.

A standardised generic protocol has been implemented at all sites. There are three sampling strategies:
- A healthcare facility-based survey of patients presenting with febrile illness with and without malaria.
- A cross sectional survey of residents of the local community served by the healthcare facility.
- A mixed methods study of local community members and healthcare providers.
In the mixed methods study, qualitative and quantitative surveys gather information on the perceptions of malaria. Including participants perceptions of malaria, radical cure, health seeking behavior, G6PD testing, and adherence to malaria treatment.
The surveys are being conducted in several countries/sites across the Asia Pacific Malaria Elimination Network. Data are being collected in a standardized format so that they can be analyzed for each site and also pooled for combined analyses. Whilst a similar protocol is used at each site, it is tailored to the local needs and burden of malaria. Not all sites have implemented all four components, depending on local need and logistical constraints.
Blood Sampling
Blood is collected by either venipuncture or finger prick depending on the participants malaria status and age. All samples are tested for malaria and G6PD deficiency using standard as well as novel diagnostics.
A selection of the blood samples are also processed for:
- Parasite genotyping
- Host genotyping (G6PD variants)
- Serological responses to malaria antigens.
Participants diagnosed with malaria are treated according to national treatment guidelines.
Project dates:
The project commenced in June 2018 and is ongoing.
Left: Workshop with the collaborators in North Sumatra, Indonesia (the V-sign is the national symbol of the fight against malaria).
Right: Sample collection in Boking Island, Indonesia.


Chinese collaborators from NIPD during laboratory training.

The ACROSS surveys were designed in consultation with country partners at the Asia Pacific Malaria Elimination Network (APMEN).
Following an open call to partners in APMEN countries, there were 10 applications. Each application was reviewed by a panel of experts from the APMEN vivax working group, chaired by Dr Effie Espino, the Director of APMEN. Selection criteria for successful applicants were the absence of former data on the topic, track record of the main applicant, local infrastructure available and a reasonable site-specific budget.
In the initial round seven applications were selected from: Bangladesh, China, India, Indonesia, Laos, Nepal, and Vietnam. With additional funding four further sites in Indonesia, Pakistan, and Bhutan were also included. As further funding becomes available, and other endemic countries request support, it is hoped that further sites will join the study.
Study sites
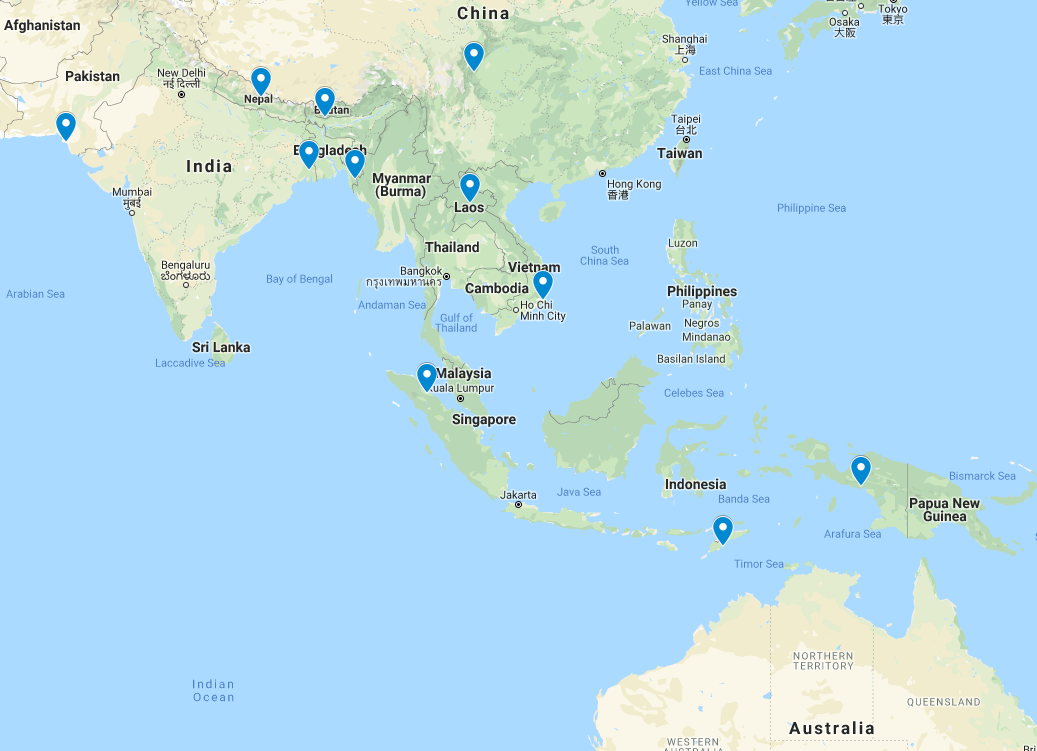
Field site in Nepal, close to Dhangadhi

Sample collection in the Chittagong Hill Tracts, Bangladesh




