Aims:
- To determine if simple, cheap regimens of oral antibiotics will lead to equivalent cure rates of impetigo (skin sores) to those achieved with penicillin injections.
- To examine the relative contribution of Staphylococcus aureus to the development of impetigo.
Objectives:
- To determine whether children, aged 12 weeks to less than 13 years, who have been diagnosed with mild or severe impetigo have non-inferior cure rates at day seven when treated with oral trimethoprim-sulfamethoxazole compared to those treated with a single dose of intramuscular benzathine penicillin G (the current standard treatment).
Summary:
We recruited 508 children with impetigo (skin sores) into an open-label, non-inferiority randomised controlled trial (RCT) involving three groups, with blinding of the outcome assessment.
Group one received a single dose of intramuscular benzathine penicillin G (Standard treatment); Group two received once-daily oral trimethoprim-sulfamethoxazole administered for five days (Alternative treatment 1); Group three received twice-daily oral trimethoprim-sulfamethoxazole administered for three days (Alternative treatment 2).
Digital photographs were taken of all of the sores and were used to determine treatment success. We compared the proportion of children within each of the respective groups who were defined as being “successfully treated” on day seven compared to baseline.
This trial is the next step in hopefully providing better treatments for skin sores.
Implications for policy and practice:
If we can develop a better alternative treatment to penicillin injections, then we will be able to recommend a regimen that will be well tolerated and lead to good adherence for an important health problem. We hope to see more kids getting treated for sores and fewer complications of sores such as kidney and heart disease.
Our research has found:
That either of the short courses of oral antibiotics work just as well as the standard treatment with penicillin injections. This is great news for kids in remote communities needing treatment for their sores, as the oral antibiotic regimen is simple, short, tastes good and is pain-free.
Chief investigators:
- Professor Jonathan Carapetis (former Menzies Director)
- Associate Professor Ross Andrews
- Professor Bart Currie
- Dr Steven Tong
- Dr Asha Bowen
Project manager:
- Irene O'Meara
Contact information:
- Irene O'Meara
Project dates:
The project commenced in 2008 and was completed in 2014.
Funders:
- National Health and Medical Research Council (NHRMC).
-
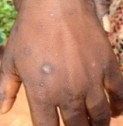
Pioneering skin sores research to improve health of children globally
A NT-based research project investigating alternative and more practical treatments for skin sores is set to benefit the millions of children worldwide who suffer from this infection.
- Bowen, A.C., Tong S.Y.C., Andrews, R.M., O’Meara, I.M, McDonald, M.I., Chatfield, M.D., Currie B.J., & Carapetis, J.R. (2014). Short course oral co-trimoxazole versus intramuscular benzathine benzylpenicillin for impetigo in a highly endemic region: an open-label, randomised, controlled, non-inferiority trial. The Lancet, early online publication 27 August 2014. doi:10.1016/S0140-6736(14)60841-2
- Bowen, A.C., Lilliebridge, R.A., Tong, S.Y., Baird, R.W., Ward, P., McDonald, M.I., et al. (2012). Is Streptococcus pyogenes Resistant or Susceptible to Trimethoprim-Sulfamethoxazole? Journal of Clinical Microbiology, 50(12),4067-72.
- Bowen, A.C., Tong, S.Y., Carapetis, J.R. (2013). Reply to "Susceptibility of Streptococcus pyogenes to trimethoprim-sulfamethoxazole". Journal of Clinical Microbiology, 51(4),1351.
- Bowen, A.C., Tong, S.Y., Chatfield, M.D., Andrews, R.M., Carapetis, J.R. (2013). Comparison of three methods for the recovery of skin pathogens from impetigo swabs collected in a remote community of Northern Territory, Australia. Royal Society of Tropical Medicine and Hygiene, 107(6), 384-9.
- Tong, S.Y., Andrews, R.M., Kearns, T., Gundjirryirr, R., McDonald, M.I., Currie, .B.J, et al. (2010). Trimethopim-sulfamethoxazole compared with benzathine penicillin for treatment of impetigo in Aboriginal children: a pilot randomised controlled trial. Journal of Paediatrics and Child Health, 46(3),131-3.

